
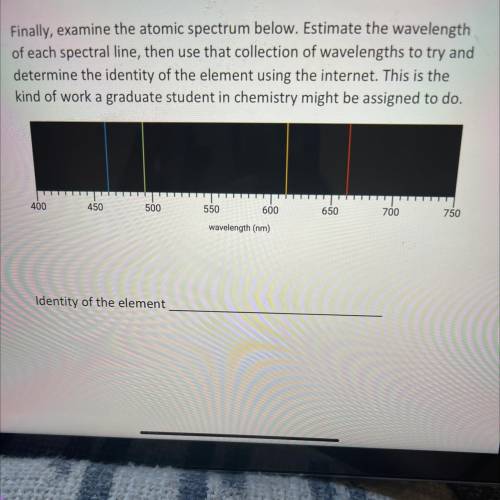
Finally, examine the atomic spectrum below. Estimate the wavelength
of each spectral line, then use that collection of wavelengths to try and
determine the identity of the element using the internet. This is the
kind of work a graduate student in chemistry might be assigned to do.
400
450
500
650
700
750
550 600
wavelength (nm)
Identity of the element

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 02:40
How many liters of hydrogen gas will be produced at stp from the reaction of 7.179×10^23 atoms of magnesium with 54.219g of phosphoric acid (h3po4) the equation is 3mg + 2h3(> mg(po4)2+3h2
Answers: 1

Chemistry, 22.06.2019 03:30
What is the relationship of air masses and the temperature of oceans?
Answers: 1

Chemistry, 22.06.2019 04:00
The rules of engagement (roe) working group is often used to (select all that apply.)
Answers: 2

Chemistry, 22.06.2019 08:00
An electron moved from shell n = 2 to shell n = 1. what most likely happened during the transition? a fraction of a photon was added. a photon of energy was absorbed. a fraction of a photon was removed. a photon of energy was released.
Answers: 1
You know the right answer?
Finally, examine the atomic spectrum below. Estimate the wavelength
of each spectral line, then us...
Questions


Mathematics, 15.02.2021 01:00

Mathematics, 15.02.2021 01:00



Computers and Technology, 15.02.2021 01:00


Mathematics, 15.02.2021 01:00

Mathematics, 15.02.2021 01:00

Arts, 15.02.2021 01:00







Mathematics, 15.02.2021 01:00


Mathematics, 15.02.2021 01:00

Mathematics, 15.02.2021 01:00



