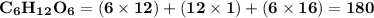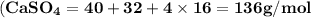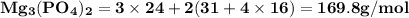Use the periodic table to calculate the molar mass of each compound below. give your answer to the correct number of significant figures. sodium hydroxide (naoh): 2 g/mol water (h2o): 4 g/mol glucose (c6h12o6): 2 g/mol calcium sulfate (caso4): 1 g/mol magnesium phosphate (mg3(po4)2): 1 g/mol

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 14:00
What mass of natural gas (ch4) must you burn to emit 276 kj of heat?
Answers: 1

Chemistry, 22.06.2019 17:30
Oil rich countries in the middle east cover about 4% of earths total land area but prossess about 48% of the worlds known oil reserves what is the main reason for high concentration of reserves in this part of the world
Answers: 3

Chemistry, 22.06.2019 19:40
Scientists have developed an explanation of a phenomenon from several verified hypotheses. the explanation has been confirmed through numerous experimental tests.which option best describes this explanation? a. scientific lawb. research questionc. hypothesisd. scientific theory
Answers: 3

Chemistry, 22.06.2019 22:00
The diagrams to the right show the distribution and arrangement of gas particles in two different containers. according to kinetic-molecular theory, which of the following statements is true? check all that apply. if the temperatures of both containers are equal, container a has greater pressure than container b. if the volume of container a decreased, its pressure would decrease. if the pressure in both containers is equal, container a has a lower temperature than container b.
Answers: 2
You know the right answer?
Use the periodic table to calculate the molar mass of each compound below. give your answer to the c...
Questions



English, 26.08.2019 18:30

Mathematics, 26.08.2019 18:30

Mathematics, 26.08.2019 18:30


Mathematics, 26.08.2019 18:30


Mathematics, 26.08.2019 18:30

History, 26.08.2019 18:30

Mathematics, 26.08.2019 18:30


Mathematics, 26.08.2019 18:30

English, 26.08.2019 18:30




Biology, 26.08.2019 18:30


Computers and Technology, 26.08.2019 18:30

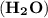 = (2 x 1) + 16 = 18 g/mol
= (2 x 1) + 16 = 18 g/mol