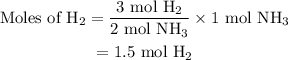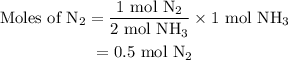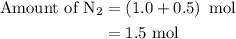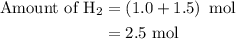Chemistry, 03.07.2019 16:30 campbell387
Amixture of n2(g) and h2(g) reacts in a closed container to form ammonia, nh3(g). the reaction ceases before either reactant has been totally consumed. at this stage 1.0 mol n2, 1.0 mol h2, and 1.0 mol nh3 are present. part a how many moles of n2 and h2 were present originally?

Answers: 1


Another question on Chemistry


Chemistry, 22.06.2019 14:40
Pastoral farming is best described as a. a method of raising livestock and moving herds b. an african method of agriculture c. a method of cultivating crops on poor soils d. a common method of desert farming select the best answer from the choices provided a b c d
Answers: 2


Chemistry, 22.06.2019 17:10
Acalorimeter is to be calibrated: 51.203 g of water at 55.2 degree c is added to a calorimeter containing 49.783 g of water at 23.5c. after stirring and waiting for the system to equilibrate, the final temperature reached is 37.6 degree c. specific heat capacity of water (s = 4.18 j/g∙degree c). calculate the calorimeter constant. (smδt)warm water = -[(smδt)cold water + (calorimeterδtcold water)]
Answers: 2
You know the right answer?
Amixture of n2(g) and h2(g) reacts in a closed container to form ammonia, nh3(g). the reaction cease...
Questions

History, 14.07.2019 07:10



Mathematics, 14.07.2019 07:10


History, 14.07.2019 07:10


Business, 14.07.2019 07:10

Advanced Placement (AP), 14.07.2019 07:10

Mathematics, 14.07.2019 07:10

Social Studies, 14.07.2019 07:10


Mathematics, 14.07.2019 07:10



Mathematics, 14.07.2019 07:10

Computers and Technology, 14.07.2019 07:10


History, 14.07.2019 07:10

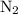 initially taken is
initially taken is 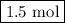 and the amount of
and the amount of 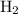 initially taken is
initially taken is 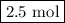 .
.
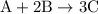
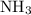 is as follows:
is as follows:
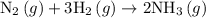
 .
.