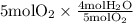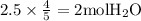Chemistry, 04.07.2019 21:50 Jasmine3864
The combustion of propane (c3h8) produces co2 and h2o: c3h8 (g) + 5o2 (g) → 3co2 (g) + 4h2o (g) the reaction of 2.5 mol of o2 with 4.6 mol of c3h8 will produce mol of h2o.

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 22:30
Using the periodic table, complete the table to describe each atom. type in your answers.a ? b? c? d? e? f?
Answers: 1

Chemistry, 22.06.2019 02:30
When you perform this reaction, what could remain at the end of the reaction? check all that apply. excess reactant aqueous copper chloride excess reactant aluminum oxygen product solid copper carbon dioxide product aqueous aluminum chloride water
Answers: 2

Chemistry, 22.06.2019 05:30
What is the mass defect of a mole of nuclei with 1.8 x 10^15 j/mol binding energy?
Answers: 1

You know the right answer?
The combustion of propane (c3h8) produces co2 and h2o: c3h8 (g) + 5o2 (g) → 3co2 (g) + 4h2o (g) the...
Questions


Mathematics, 11.07.2019 22:40



History, 11.07.2019 22:40




History, 11.07.2019 22:40


Mathematics, 11.07.2019 22:40


English, 11.07.2019 22:40



English, 11.07.2019 22:40


Mathematics, 11.07.2019 22:40


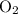 with 4.6 mol
with 4.6 mol 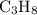 will produce 2mol
will produce 2mol 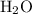 .
.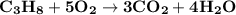
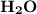 produced will be,
produced will be,