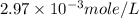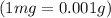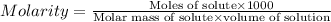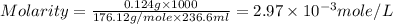Chemistry, 05.07.2019 00:40 ayoismeisalex
One cup of fresh orange juice contains 124 mg of ascorbic acid (vitamin c, c6h8o6). given that one cup = 236.6 ml, calculate the molarity of vitamin c in organic juice.

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 20:10
Why is the vapor pressure of a warm lake higher than the vapor pressure of a cold lake? o a. warm water has a greater heat of vaporization. ob. warm water evaporates more quickly. cool water evaporates more quickly. od. cool water has a greater heat of vaporization.
Answers: 1

Chemistry, 21.06.2019 22:30
What are the charges of the subatomic particles by choosing the answer from the drop down menu. protons have a (+1,0,or-1). (protons,neutrons,electrons) have a 0 charge. 3.) electrons have a (+1,0,-1)
Answers: 2

Chemistry, 22.06.2019 01:50
7. what temperature is need to just dissolve 50 g of nh4cl in 75 g of water? '
Answers: 1

Chemistry, 22.06.2019 02:10
When 225mg of anthracene, c14h10(s), was burned in a bomb calorimeter the temperature rose by 1.75k. calculate the calorimeter constant. by how much will the temperature rise when 125mg of phenol, c6h5oh(s), is burned in the calorimeter under the same conditions? (δch< (c14h10,s)=–7061 kj mol−1.)
Answers: 3
You know the right answer?
One cup of fresh orange juice contains 124 mg of ascorbic acid (vitamin c, c6h8o6). given that one c...
Questions





Mathematics, 27.01.2020 05:31





English, 27.01.2020 05:31



Chemistry, 27.01.2020 05:31

History, 27.01.2020 05:31

English, 27.01.2020 05:31


Social Studies, 27.01.2020 05:31


Geography, 27.01.2020 05:31