Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 15:30
The isotonic saline solution described in part a is connected to an unknown solution via a semipermeable membrane, the unknown solution level drops. based on this information, what can be said about these two solutions?
Answers: 1

Chemistry, 22.06.2019 18:00
Answer asap need it by wednesday morning carry out the following calculations on ph and ka of from data. i. calculate the ph of 0.02m hcl ii. calculate the ph of 0.036m naoh iii. calculate the ph of 0.36m ca(oh)2 iv. calculate the ph of 0.16m ch3cooh which has ka = 1.74 x 10-5 mol dm-3 v. calculate ka for weak acid ha which has a ph of 3.65 at 0.30m concentration vi. calculate the ka of a solution made by mixing 15.0 cm3 0.2m ha and 60.0 cm3 0.31m a-. [ph= 3.80] vii. calculate the ph of a solution made by mixing 15.0 cm3 0.1m naoh and 35.0 cm3 0.2m hcooh. [ka = 1.82 x 10-4 m]
Answers: 1

Chemistry, 23.06.2019 03:00
What volume does 1.70 ×10–3 mol of chlorine gas occupy if its temperature is 20.2 °c and its pressure is 795 mm hg?
Answers: 3

Chemistry, 23.06.2019 04:31
One student said that the investigation was not valid (a fair test). write a plan for the investigation that includes improvements to the method and apparatus
Answers: 1
You know the right answer?
Calculate the χacetone and χcyclohexane in the vapor above the solution. p°acetone = 229.5 torr and...
Questions

Business, 07.01.2021 20:30


English, 07.01.2021 20:30

Mathematics, 07.01.2021 20:30

Mathematics, 07.01.2021 20:30

Arts, 07.01.2021 20:30

Mathematics, 07.01.2021 20:30


History, 07.01.2021 20:30



Mathematics, 07.01.2021 20:30

Mathematics, 07.01.2021 20:30

Social Studies, 07.01.2021 20:30


Mathematics, 07.01.2021 20:30


Mathematics, 07.01.2021 20:30


Health, 07.01.2021 20:30

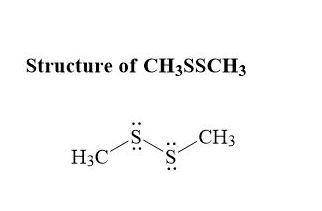
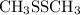 is shown in the attached image.
is shown in the attached image.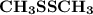 (Refer to the structure in the attached image):
(Refer to the structure in the attached image):![\begin{aligned} {\text{Total valence electrons}}\left( {{\text{TVE}}} \right) &= \left[ {\left( {\text{2}} \right)\left( {\text{4}} \right) + \left( {\text{2}} \right)\left( {\text{6}} \right) + \left( 6 \right)\left( 1 \right)} \right] \\ & = 26 \\ \end{aligned}](/tpl/images/0071/6883/4c737.png)