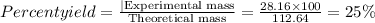Chemistry, 12.07.2019 21:40 jsharma57p7enrw
Give the percent yield when 28.16 g of co2 are formed from the reaction of 8.000 moles of c8h18 with 4.000 moles of o2. 2 c8h18 + 25 o2 → 16 co2 + 18 h2o

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 14:00
What is the molar mass of babr2? a. 217.2 g/mol b. 297.1 g/mol c. 354.5 g/mol d. 434.4 g/mol
Answers: 1

Chemistry, 22.06.2019 02:00
What is the maximum number of electrons that an atomic orbital can contain?
Answers: 1

Chemistry, 22.06.2019 09:10
When a nucleus absorbs a neutron and then breaks apart, there are many products of the reaction. what is not a product of a nuclear fission reaction
Answers: 1

Chemistry, 22.06.2019 09:30
What are scientists who study fossils called? ( a ) astronomers. ( b ) biologists. ( c ) geologists. ( d ) paleontologists.
Answers: 2
You know the right answer?
Give the percent yield when 28.16 g of co2 are formed from the reaction of 8.000 moles of c8h18 with...
Questions


History, 04.08.2019 06:30



Social Studies, 04.08.2019 06:30


Biology, 04.08.2019 06:30

Mathematics, 04.08.2019 06:30


Spanish, 04.08.2019 06:30

Social Studies, 04.08.2019 06:30


English, 04.08.2019 06:30

Health, 04.08.2019 06:30



History, 04.08.2019 06:30

Spanish, 04.08.2019 06:30


Spanish, 04.08.2019 06:30

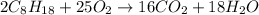
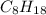 , then 4 moles of oxygen will react with
, then 4 moles of oxygen will react with 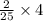 moles of
moles of 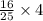 moles of carbon-dioxide gas that is 2.56 moles.
moles of carbon-dioxide gas that is 2.56 moles.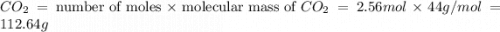
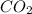 = 28.16 g
= 28.16 g