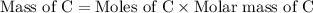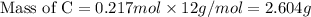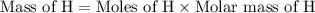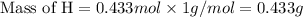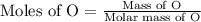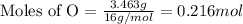Chemistry, 14.07.2019 20:50 live4dramaoy0yf9
If 6.50 g of the unknown compound contained 0.217 mol of c and 0.433 mol of h , how many moles of oxygen, o , were in the sample?

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 01:20
1. suppose a reaction mixture, when diluted with water, afforded 300 ml of an aqueous solution of 30 g of the reaction product malononitrile [ch2(cn)2], which is to be isolated by extraction with ether. the solubility of malononitrile in ether at room temperature is 20.0 g/100 ml, and in water is 13.3 g/100 ml. what weight of malononitrile would be recovered by extraction with (a) three 100-ml portions of ether and (b) one 300-ml portion of ether? suggestion: for each extraction, let x equal the weight extracted into the ether layer. in part (a), the concentration in the ether layer is x/100 and in the water layer is (30 x)/300; the ratio of these quantities is equal to k 20/13.3.
Answers: 2

Chemistry, 22.06.2019 10:30
Astudent reacts 13 moles of iron with 21 moles of oxygen according to the following equation:
Answers: 1

Chemistry, 22.06.2019 15:00
Which substance is a steroid? cholesterol fatty acid monosaccharide trans fat
Answers: 1

Chemistry, 22.06.2019 18:30
The famous scientist galileo galilei did several experiments with sloping planes, which he rolled metal balls down so that he could study motion. by changing the slope, he could study how the speed at which the ball rolled was affected. what was the independent variable in galileo's experiment? a. the speed of the ball b. the slope of the plane c. whether the ball moved d. what the ball was made of
Answers: 2
You know the right answer?
If 6.50 g of the unknown compound contained 0.217 mol of c and 0.433 mol of h , how many moles of ox...
Questions


Mathematics, 02.03.2021 20:10


Physics, 02.03.2021 20:10



Mathematics, 02.03.2021 20:10

Mathematics, 02.03.2021 20:10


Biology, 02.03.2021 20:10

Mathematics, 02.03.2021 20:10

Mathematics, 02.03.2021 20:10

Mathematics, 02.03.2021 20:10