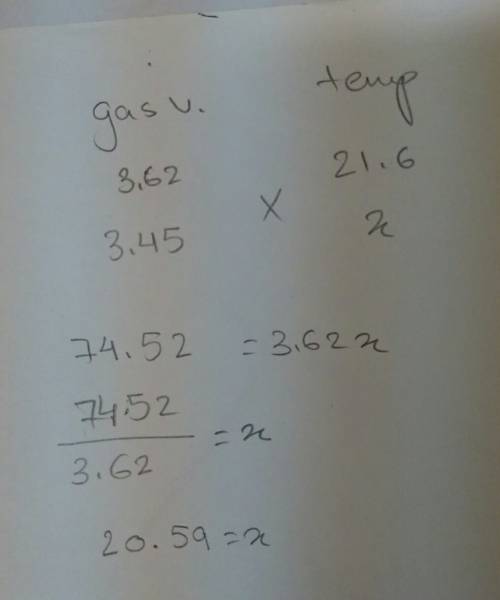Chemistry, 19.07.2019 08:30 Britny2386
Agas sample is held at constant pressure. the gas occupies 3.62 l of volume when the temperature is 21.6°c. determine the temperature at which the volume of the gas is 3.45 l.

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 14:00
How does the presence of oxygen affect the chemical pathways used to extract energy from glucose?
Answers: 3

Chemistry, 22.06.2019 18:00
Answer asap need to be answered by wednesday morning explain how a buffer works, using an ethanoic acid / sodium ethanoate system including how the system resists changes in ph upon addition of a small amount of base and upon addition of a small amount of acid respectively. include the following calculations in your i. calculate the ph of a solution made by mixing 25cm3 0.1m ch3cooh and 40cm3 0.1m ch3coo-na+. [ka = 1.74 x 10-5 m] ii. calculate the ph following the addition of a 10cm3 portion of 0.08 m naoh to 500cm3 of this buffer solution. iii. calculate the ph following the addition of a 10cm3 portion of 0.08 m hcl to 200cm3 of the original buffer solution.
Answers: 3

Chemistry, 22.06.2019 22:40
Percent ionization for a weak acid (ha) is determined by the following formula: percent ionization=[ha] ionized[ha] initial×100%for strong acids, ionization is nearly complete (100%) at most concentrations. however, for weak acids, the percent ionization changes significantly with concentration. the more diluted the acid is, the greater percent ionization.a certain weak acid, ha, has a ka value of 9.4×10? 7.part acalculate the percent ionization of ha in a 0.10 m solution.part bcalculate the percent ionization of ha in a 0.010 m solution
Answers: 1

Chemistry, 23.06.2019 02:00
Calculate the molarity of each aqueous solution: a. 78.0 ml of 0.240 m naoh diluted to 0.250 l with water b. 38.5 ml of 1.2 m hno3 diluted to 0.130 l with water
Answers: 1
You know the right answer?
Agas sample is held at constant pressure. the gas occupies 3.62 l of volume when the temperature is...
Questions


Computers and Technology, 05.02.2021 14:00

Advanced Placement (AP), 05.02.2021 14:00

Physics, 05.02.2021 14:00

Mathematics, 05.02.2021 14:00


Mathematics, 05.02.2021 14:00

Mathematics, 05.02.2021 14:00

Mathematics, 05.02.2021 14:00


Mathematics, 05.02.2021 14:00

Mathematics, 05.02.2021 14:00

Biology, 05.02.2021 14:00

Mathematics, 05.02.2021 14:00


Mathematics, 05.02.2021 14:00

Mathematics, 05.02.2021 14:00


Mathematics, 05.02.2021 14:00