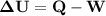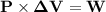An ideal gaseous reaction (which is a hypothetical gaseous reaction that conforms to the laws governing gas behavior) occurs at a constant pressure of 45.0 atm and releases 68.0 kj of heat. before the reaction, the volume of the system was 6.40 l . after the reaction, the volume of the system was 2.80 l . calculate the total internal energy change, δe, in kilojoules.

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 07:00
Achemist wants to extract copper metal from copper chloride solution. the chemist places 0.50 grams of aluminum foil in a solution containing 0.75 grams of copper (ii) chloride. a single replacement reaction takes place. (ii) chloride. a single replacement reaction takes place. which statement explains the maximum amount of copper that the chemist can extract using this reaction? a) approximately 0.36 grams, because copper (ii) chloride acts as a limiting reactant b) approximately 1.8 grams, because copper (ii) chloride acts as a limiting reactant c) approximately 0.36 grams, because aluminum acts as a limiting reactant d) approximately 1.8 grams, because aluminum acts as a limiting reactant
Answers: 3

Chemistry, 22.06.2019 22:30
Which of these statements best explains why space exploration should be encouraged? it prepares humans to live without oxygen. it dispel myths about objects in space. it prevents comets and asteroids from striking earth. it creates technology to absorb harmful radiations in space.
Answers: 1


Chemistry, 23.06.2019 01:00
The time that is taken by neptune once around the sun is called
Answers: 1
You know the right answer?
An ideal gaseous reaction (which is a hypothetical gaseous reaction that conforms to the laws govern...
Questions

History, 29.07.2019 00:00

Business, 29.07.2019 00:00




History, 29.07.2019 00:00


English, 29.07.2019 00:00


English, 29.07.2019 00:00


Social Studies, 29.07.2019 00:00


English, 29.07.2019 00:00

Computers and Technology, 29.07.2019 00:00


Social Studies, 29.07.2019 00:00