
Chemistry, 29.07.2019 19:00 elitehairnerd1964
Which of the following best explains the polarity of water (h2o)? a. protons are more attracted to the hydrogen atoms, so the hydrogen atoms become positively charged. b. since the oxygen atom has more electrons than the hydrogen atoms, it is negatively charged. c. electrons are more attracted to the oxygen atom, so there is a partial negative charge on the oxygen atom. d. since there are two positive hydrogen atoms and only one negative oxygen atom, the water molecule is positively charged.

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 17:10
There are 6.022 x 10^23 atoms of hg in 1 mole of hg. the number of atoms in 4.5 moles of hg can be found by multiplying 4.5 by 6.022 x 10^23 a. 2.7 x 10^24 b. 27 x 10^23 c. 2.71 x10^24 d. 27.099 x 10^23
Answers: 3


Chemistry, 22.06.2019 04:30
In which phase(s) do the molecules take the shape of the container?
Answers: 1

Chemistry, 22.06.2019 05:50
What are transitions between a liquid and gas called? identify which way they are transitioning
Answers: 2
You know the right answer?
Which of the following best explains the polarity of water (h2o)? a. protons are more a...
Questions

Mathematics, 15.09.2020 21:01

Mathematics, 15.09.2020 21:01

Mathematics, 15.09.2020 21:01

Mathematics, 15.09.2020 21:01

Mathematics, 15.09.2020 21:01

Mathematics, 15.09.2020 21:01

Social Studies, 15.09.2020 21:01

Mathematics, 15.09.2020 21:01

Mathematics, 15.09.2020 21:01

English, 15.09.2020 21:01

Mathematics, 15.09.2020 21:01

Mathematics, 15.09.2020 21:01

English, 15.09.2020 21:01

Mathematics, 15.09.2020 21:01

English, 15.09.2020 21:01

Mathematics, 15.09.2020 21:01

Mathematics, 15.09.2020 21:01

Mathematics, 15.09.2020 21:01

Mathematics, 15.09.2020 21:01

Mathematics, 15.09.2020 21:01

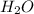 . This shows that there are two hydrogen atoms and one oxygen atom.
. This shows that there are two hydrogen atoms and one oxygen atom. 

