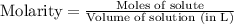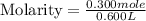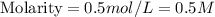Chemistry, 05.08.2019 00:30 Ezekielcassese
Calculate the molarity of a solution that contains 0.300 moles of csbr in 0.600 l of water.

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 22:30
How many moles are in 250 grams of tungsten (w)? * 4.4x10^23 moles 4.2x10^23 moles 0.7 moles 1.4 moles
Answers: 3

Chemistry, 22.06.2019 03:30
Select the correct answer. when carbon dioxide dissolves in water, it sometimes reacts with water to form carbonic acid as in this balanced equation: co2 + h2o → h2co3. if 495 milliliters of carbon dioxide at 25°c and 101.3 kilopascals reacts with excess water, what is the theoretical yield of carbonic acid? use the periodic table and the ideal gas resource a. 0.889 g b. 1.10g c. 1.27g d. 2.02g what's the answer! quick!
Answers: 1

Chemistry, 22.06.2019 14:40
Choose an equation that represents an enzyme-catalyzed reaction. (a) enzyme + substrate → enzyme-substrate complex (b) enzyme + substrate ←−→ enzyme + products (c) enzyme + substrate ←−→ enzyme-substrate complex → enzyme + products (d) enzyme + substrate ←−→ enzyme-substrate complex → enzyme-substrate complex + products
Answers: 2

Chemistry, 22.06.2019 18:00
Hydrogenation reactions, in which h2 and an "unsaturated" organic compound combine, are used in the food, fuel, and polymer industries. in the simplest case, ethene (c2h4) and h2 form ethane (c2h6). if 140 kj is given off per mole of c2h4 reacting, how much heat (in mj) is released when 12 kg of c2h6 forms?
Answers: 2
You know the right answer?
Calculate the molarity of a solution that contains 0.300 moles of csbr in 0.600 l of water....
Questions

Mathematics, 27.08.2020 02:01


Computers and Technology, 27.08.2020 02:01







Computers and Technology, 27.08.2020 02:01


Biology, 27.08.2020 02:01


Mathematics, 27.08.2020 02:01


Biology, 27.08.2020 02:01


Mathematics, 27.08.2020 02:01

Mathematics, 27.08.2020 02:01

Mathematics, 27.08.2020 02:01