
Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 07:20
Describing intermolecular forces use the drop down menus to match the type of intermolecular force to its name dipole dipole interactions dipole induced dipole interactions london dispersion forces hydrogen bond van der waals forces done
Answers: 1

Chemistry, 22.06.2019 10:00
In a water molecule, hydrogen and oxygen are held together by a(an) bond. a) double covalent b) ionic c) nonpolar covalent d) hydrogen e) polar covalent
Answers: 1


Chemistry, 22.06.2019 22:30
You just calculated that the heat of fusion for chloromethane is 6400 j/mol. the heat of fusion for hydrogen is 120 j/mol.? which of the following account for this difference? more than one correcta. chloromethane can absorb more energy at the same temperature. b. hydrogen has stronger intermolecular forces than chloromethane. c. hydrogen molecules can pack more closely than chloromethane molecules. d. chloromethane experiences dipole-dipole interactions. e. chloromethane has a higher molar mass than hydrogen.
Answers: 3
You know the right answer?
Do all molecules contain only single bonds...
Questions






Health, 15.10.2020 20:01

Mathematics, 15.10.2020 20:01


Mathematics, 15.10.2020 20:01

Mathematics, 15.10.2020 20:01

History, 15.10.2020 20:01

Biology, 15.10.2020 20:01

Spanish, 15.10.2020 20:01



Biology, 15.10.2020 20:01

History, 15.10.2020 20:01



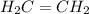 is ethene molecule and it contains a double bond.
is ethene molecule and it contains a double bond.

