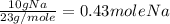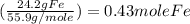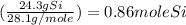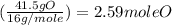Chemistry, 31.07.2019 18:00 pankajyadav7402
If a laboratory analysis of the mineral aegirine yields 10.0% na, 24.2% fe, 24.3% si, and 41.5% o, what is the empirical formula of the compound?

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 12:00
What is a possible quantum number set for an electron in the 3s orbital of a magnesium atom
Answers: 1

Chemistry, 22.06.2019 14:30
100 grams of molten lead (600°c) is used to make musket balls. if the lead shot is allowed to cool to room temperature (21°c), what is the change in entropy (in j/k) of the lead? (for the specific heat of molten and solid lead use 1.29 j/g⋅°c; the latent heat of fusion and the melting point of lead are 2.45 × 104 j/kg and 327°c, respectively.)
Answers: 1

Chemistry, 23.06.2019 00:30
Maya wrote if you step to describe how carbon circulates between the atmosphere and living organisms
Answers: 1

Chemistry, 23.06.2019 00:30
Element j is 1s 2s 2p 3s . (i) how many unpaired electrons does j have? (ii) is j a good oxidizing agent or a reducing agent? (iii) state reason for the answer.
Answers: 1
You know the right answer?
If a laboratory analysis of the mineral aegirine yields 10.0% na, 24.2% fe, 24.3% si, and 41.5% o, w...
Questions

Chemistry, 20.11.2020 20:40


Biology, 20.11.2020 20:40


Physics, 20.11.2020 20:40


Mathematics, 20.11.2020 20:40

History, 20.11.2020 20:40

History, 20.11.2020 20:40

Engineering, 20.11.2020 20:40

Mathematics, 20.11.2020 20:40


Mathematics, 20.11.2020 20:40



Mathematics, 20.11.2020 20:40

Mathematics, 20.11.2020 20:40

Mathematics, 20.11.2020 20:40