What is the expected oxidation state for the most common ion of element 2
...

Chemistry, 24.09.2019 23:30 jaliyahrobinson1
What is the expected oxidation state for the most common ion of element 2
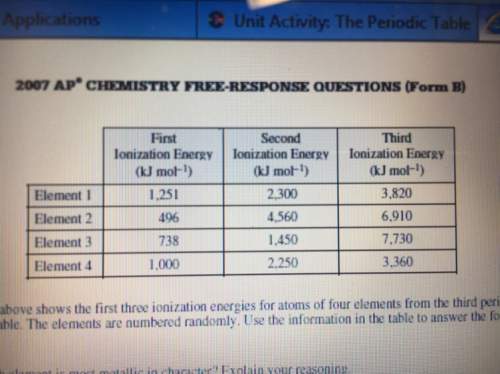

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 23:30
Agroup of students is studying convection currents. they fill two identical balloons with the same amount of helium. one balloon is placed in a freezer and the other in an area with warm air. after 10 minutes, the balloons are released from a height of 1 meter. which of the following do the students most likely observe? a. the balloons both rise. the cold balloon is larger than the warm balloon. b. the balloons rise at the same rate. both balloons are the same size. c. the warm balloon expands and rises. the cold balloon shrinks and sinks. d. the cold balloon expands and rises. the warm balloon shrinks and sinks.
Answers: 2

Chemistry, 22.06.2019 00:30
What is the most stable monatomic ion formed from nitrogen
Answers: 2

Chemistry, 22.06.2019 18:50
Question 3(multiple choice worth 4 points) (04.04 lc) what does it mean when an element is reduced? it empties a valance shell, reducing its atomic radius. it gains electrons, reducing its overall charge. it increases electronegativity, reducing its ability to bond. it loses electrons, reducing its electron number.
Answers: 1

Chemistry, 22.06.2019 19:30
Phosphorous can form an ion called phosphide, which has the formula p3−. this ion can form an ion called phosphide, which has the formula p3−. this ion properties very similar to those of pforms when a phosphorus atom loses three protonsis called a cationcontains 18 electrons
Answers: 2
You know the right answer?
Questions


Mathematics, 11.07.2019 02:50


History, 11.07.2019 02:50

History, 11.07.2019 02:50

Biology, 11.07.2019 02:50

Social Studies, 11.07.2019 02:50

Biology, 11.07.2019 02:50

Biology, 11.07.2019 02:50

Biology, 11.07.2019 02:50




History, 11.07.2019 03:00


Health, 11.07.2019 03:00

Mathematics, 11.07.2019 03:00





