

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 02:40
Consider the nuclear equation below. 239/94 pu—-> x+ 4/2 he. what is x?
Answers: 2


Chemistry, 23.06.2019 17:00
Using this reversible reaction, answer the questions below: n2o4 2no2 (colorless) (reddish-brown) -as the temperature increased, what happened to the n2o4 concentration? -was the formation of reactants or products favored by the addition of heat? -which reaction is exothermic? right to left or left to right? -if the change of enthalpy of this reaction when proceeding left to right is 14 kcal, which chemical equation is correct? n2o4 2no2 + 14 kcal n2o4 2no2, hr = +14 kcal n2o4 + 14 kcal 2no2 n2o4 2no2, hr = -14 kcal
Answers: 1

Chemistry, 23.06.2019 17:00
Under which law can a hotel guest sue for an injury that happened at the hotel’s spa? ]
Answers: 3
You know the right answer?
Muriatic acid is the commercial name for hydrochloric acid that can be purchased from hardware store...
Questions






Medicine, 22.02.2022 08:10


Mathematics, 22.02.2022 08:20

Advanced Placement (AP), 22.02.2022 08:20




Mathematics, 22.02.2022 08:20

Arts, 22.02.2022 08:20

Computers and Technology, 22.02.2022 08:20

Chemistry, 22.02.2022 08:20


English, 22.02.2022 08:20


Chemistry, 22.02.2022 08:20

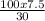 = 25 g solution
= 25 g solution

