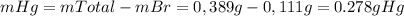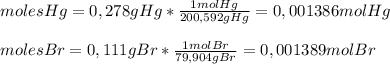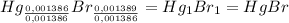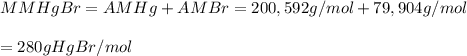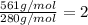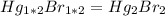Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 01:30
When an object falls through the air and encounters air resistance its overall speed will be than if it had not encountered air resistance? (one word answer)
Answers: 2

Chemistry, 22.06.2019 07:30
The volume of helium in a blimp is 6.28 x 10^9 millimeters. the density of helium in the blimp is .1786 kilogram/meter^3. find the mass of the helium in the blimp.
Answers: 1

Chemistry, 22.06.2019 21:50
If e is the symbol for an element, which two of the following symbols represent isotopes of the same element? 1. e2. e3. ea.1 and 2c.1 and 4b.3 and 4d.2 and 3
Answers: 2

Chemistry, 23.06.2019 01:00
Which statement is true regarding the diagram of circle p? the sum of y and z must be 2x. the sum of y and z must be x. the difference of z and y must be 2x. the difference of z and y must be x
Answers: 1
You know the right answer?
Asample of a compound of mercury and bromine with a mass of 0.389 g was found to contain 0.111 g bro...
Questions

Mathematics, 10.03.2021 18:10

Biology, 10.03.2021 18:10

Mathematics, 10.03.2021 18:10


Chemistry, 10.03.2021 18:10

Mathematics, 10.03.2021 18:10



Mathematics, 10.03.2021 18:10


English, 10.03.2021 18:10

Mathematics, 10.03.2021 18:10





Mathematics, 10.03.2021 18:10

Mathematics, 10.03.2021 18:10

Mathematics, 10.03.2021 18:10

Mathematics, 10.03.2021 18:10