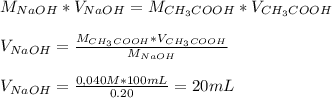Chemistry, 29.07.2019 11:30 natishtaylor1p8dirz
Calculate the volume in ml of 0.20m naoh needed to react completely with 100.ml of 0.040m acetic acid.

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 10:50
8) a mixture of he, ne and ar has a pressure of 7.85 atm. if the ne has a mole fraction of 0.47 and 8) ar has a mole fraction of 0.23, what is the pressure of he? a) 4.2 atm b) 3.7 atm c) 5.5 atm d) 2.4 atm e) 1.8 atm
Answers: 1

Chemistry, 22.06.2019 11:40
Which of these expressions are correct variations of the combined gas law? p1v1t2 = p2v2t1 both
Answers: 2

Chemistry, 22.06.2019 13:00
How many moles of sulfur dioxide are produced when 4.38 moles of oxygen completely react with iron (iv) sulfide
Answers: 2

Chemistry, 22.06.2019 14:10
13. a covalent bond between two atoms is likely to be polar if: a. one of the atoms is much more electronegative than the other. b. the two atoms are equally electronegative. c. the two atoms are of the same element. d. the bond is part of a tetrahedrally shaped molecule. e. one atom is an anion.
Answers: 1
You know the right answer?
Calculate the volume in ml of 0.20m naoh needed to react completely with 100.ml of 0.040m acetic aci...
Questions



History, 15.04.2020 15:32


Computers and Technology, 15.04.2020 15:32





Biology, 15.04.2020 15:32

Biology, 15.04.2020 15:32

Geography, 15.04.2020 15:32

Social Studies, 15.04.2020 15:33




Mathematics, 15.04.2020 15:33


Chemistry, 15.04.2020 15:33