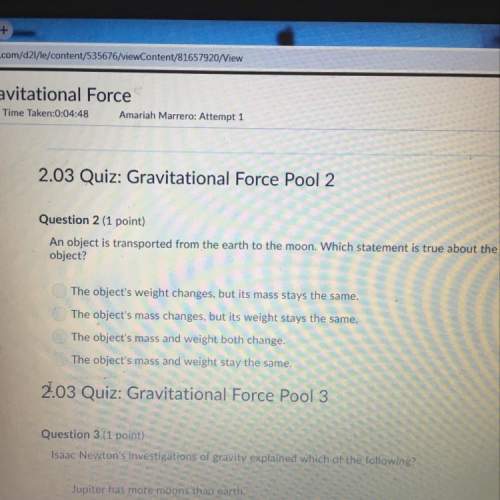
Chemistry, 29.07.2019 01:30 laqu33n021
How many moles of oxygen are produced by decomposing 40.5 g of h2o2 (molar mass = 34.0 g/mol) according to the equation: h2o2 --> h2o + o2 (unbalanced)?

Answers: 1


Another question on Chemistry


Chemistry, 22.06.2019 05:30
Match the following vocabulary terms to their definitions. 1. amount of energy required to change 1 gram of material from the solid to the liquid state at its melting point 2. a measure of the kinetic energy of the particles of a substance 3. the amount of heat energy required to raise the temperature of 1 gram of liquid water from 14.5°c to 15.5°c 4. amount of energy required to change 1 gram of material from the liquid to the gaseous state at its boiling point 5. the amount of energy required to change 1 gram of a substance 1°c a. temperature b. latent heat of vaporization c. latent heat of fusion d. calorie e. specific heat
Answers: 1

Chemistry, 22.06.2019 05:40
Consider the elements bromine and chlorine; which elements has a larger ionic radius ?
Answers: 1

You know the right answer?
How many moles of oxygen are produced by decomposing 40.5 g of h2o2 (molar mass = 34.0 g/mol) accord...
Questions

History, 14.09.2020 22:01

Mathematics, 14.09.2020 22:01

Mathematics, 14.09.2020 22:01

Mathematics, 14.09.2020 22:01

History, 14.09.2020 22:01

Mathematics, 14.09.2020 22:01

Mathematics, 14.09.2020 22:01

Mathematics, 14.09.2020 22:01

Mathematics, 14.09.2020 22:01

Mathematics, 14.09.2020 22:01

English, 14.09.2020 22:01

Mathematics, 14.09.2020 23:01

Mathematics, 14.09.2020 23:01

Mathematics, 14.09.2020 23:01

Mathematics, 14.09.2020 23:01

Social Studies, 14.09.2020 23:01

Chemistry, 14.09.2020 23:01

Social Studies, 14.09.2020 23:01

Mathematics, 14.09.2020 23:01

History, 14.09.2020 23:01