
Chemistry, 28.07.2019 14:00 vernelljordon
For the nitrogen fixation reaction, 3h2(g) + n2(g) 2nh3(g), kc = 6.0 × 10–2 at 500°c. if 0.250 m h2 and 0.050 m nh3 are present at equilibrium, what is the equilibrium concentration of n2? a. 3.3 m b. 2.7 m c. 0.20 m d. 0.083 m e. 0.058 m

Answers: 1


Another question on Chemistry




Chemistry, 22.06.2019 14:40
Choose an equation that represents an enzyme-catalyzed reaction. (a) enzyme + substrate → enzyme-substrate complex (b) enzyme + substrate ←−→ enzyme + products (c) enzyme + substrate ←−→ enzyme-substrate complex → enzyme + products (d) enzyme + substrate ←−→ enzyme-substrate complex → enzyme-substrate complex + products
Answers: 2
You know the right answer?
For the nitrogen fixation reaction, 3h2(g) + n2(g) 2nh3(g), kc = 6.0 × 10–2 at 500°c. if 0.250 m h2...
Questions

Mathematics, 23.07.2019 20:30


Social Studies, 23.07.2019 20:30



Biology, 23.07.2019 20:30

English, 23.07.2019 20:30

English, 23.07.2019 20:30

History, 23.07.2019 20:30

History, 23.07.2019 20:30



Physics, 23.07.2019 20:30

Mathematics, 23.07.2019 20:30


English, 23.07.2019 20:30




![Kc=\frac{[NH_3]^2_{eq}}{[H_2]^3_{eq}[N_2]_{eq}}](/tpl/images/0143/1009/c1580.png)
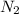 is the unique unknown, solving for it one obtains:
is the unique unknown, solving for it one obtains:![[N_2]_{eq}=\frac{[NH_3]^2_{eq}}{[H_2]^3_{eq}Kc}](/tpl/images/0143/1009/e7601.png)
![[N_2]_{eq}=\frac{(0.050M)^2}{(0.250M)^3(6.0x10^{-2})}](/tpl/images/0143/1009/c87cf.png)
![[N_2]_{eq}=2.7M](/tpl/images/0143/1009/d60cf.png)



