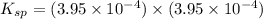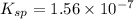Chemistry, 28.07.2019 10:30 Cartucho1978
The solubility of copper(i) chloride is 3.91 mg per 100.0 ml of solution. calculate ksp for cucl (cucl=99.00 g mol-1).

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 05:50
Calculate the number of molecules present in 0.750 mol of mgo.
Answers: 3



Chemistry, 22.06.2019 15:30
Using the first volume and temperature reading on the table as v1 and t1, solve for the unknown values in the table below. remember to use the rules of significant figures when entering your numeric response.
Answers: 2
You know the right answer?
The solubility of copper(i) chloride is 3.91 mg per 100.0 ml of solution. calculate ksp for cucl (cu...
Questions


Computers and Technology, 02.10.2019 05:10


Computers and Technology, 02.10.2019 05:10
















 is
is 
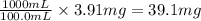 of CuCl
of CuCl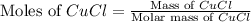
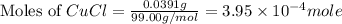
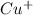 and
and 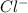 ion.
ion.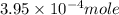
![[Cu^+]](/tpl/images/0142/5384/41578.png) =
= ![[Cl^-]](/tpl/images/0142/5384/0726e.png) =
= 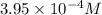
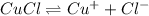
![K_{sp}=[Cu^{+}][Cl^{-}]](/tpl/images/0142/5384/fd1d1.png)