
Which characteristics of metal atoms explain why valence electrons in a metal are delocalized? metal atoms are large and have high electronegativities. metal atoms are small and have low electronegativities. metal atoms are large and have low electronegativities. metal atoms are small and have high electronegativities.

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 17:00
Why do sodium and neon have vastly different chemical and physical properties despite having similar atomic masses?
Answers: 2

Chemistry, 22.06.2019 03:30
If you have 5.25 grams of methane (ch4), how many grams of co2 will you produce ?
Answers: 1

Chemistry, 22.06.2019 14:50
Which of the following is most likely true about water in chemical solutions?
Answers: 1

Chemistry, 22.06.2019 22:30
Which one of the following bonds would you expect to be the most polar? a) b–h b) n–h c) p–h d) al–h e) c–h
Answers: 1
You know the right answer?
Which characteristics of metal atoms explain why valence electrons in a metal are delocalized? met...
Questions

Biology, 27.01.2021 21:50

Mathematics, 27.01.2021 21:50





Mathematics, 27.01.2021 21:50




Mathematics, 27.01.2021 21:50

English, 27.01.2021 21:50


Mathematics, 27.01.2021 21:50




Mathematics, 27.01.2021 21:50


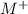 ions and group II elements are alkaline earth metals, they can give 2 electrons to form
ions and group II elements are alkaline earth metals, they can give 2 electrons to form 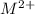 ions.
ions.

