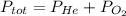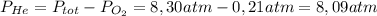He partial pressure of oxygen gas in our atmosphere is 0.21 atm. this is the partial pressure at which human lungs have evolved to be able to breathe this gas. a scuba diver, will thus still have to breath oxygen at this pressure even when diving way down in the water. if a mixture of helium and oxygen (heliox) in his tank is at a pressure of 8.30 atm, what must the partial pressure be of helium to keep the partial pressure of oxygen at 0.21 atm?

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 11:30
Which statement best describes the flow of energy in this scenario
Answers: 1

Chemistry, 22.06.2019 14:30
How do temperature and salinity affect deepwater currents? as temperatures and salinity levels of water increase, the water rises to the surface where it creates currents as it moves to colder regions. they create changes in wind direction, moving denser water in the same direction as the wind and causing the deepwater circulation patterns found in the ocean. they equalize the forces on undersea currents caused by the coriolis effect as they replace more dense water with less dense water. they create density differences that cause dense deepwater currents to flow toward the equator where they displace less dense, warmer water above them.
Answers: 2


Chemistry, 23.06.2019 09:00
Which factor is likely to impact the possible number of compounds? presence of unlimited number of elements in the periodic table the inability of atoms to align perfectly with other atoms the ability of all elements to react with every other element all elements being equally reactive
Answers: 2
You know the right answer?
He partial pressure of oxygen gas in our atmosphere is 0.21 atm. this is the partial pressure at whi...
Questions

Physics, 06.03.2020 01:00









Computers and Technology, 06.03.2020 01:00



Mathematics, 06.03.2020 01:00

Mathematics, 06.03.2020 01:00

Biology, 06.03.2020 01:00