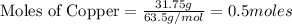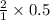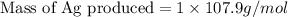Chemistry, 25.07.2019 17:30 michaellagann2020
Copper metal (cu) reacts with silver nitrate (agno3) in aqueous solution to form ag and cu(no3)2. an excess of agno3 is present. the balanced chemical equation is shown below. cu + 2agno3 mc021-1.jpg cu(no3)2 + 2ag the molar mass of cu is 63.5 g/mol. the molar mass of ag is 107.9 g/mol. what mass, in grams, of ag is produced from reaction of 31.75 g of cu? 26.95 107.9 215.91 431.82

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 09:40
In the lab, ammonia was mixed with water to form ammonium hydroxide. what is/are the reactant(s)? o water and ammonia o ammonia o ammonium hydroxide need
Answers: 2

Chemistry, 22.06.2019 14:30
In water, a strong acid will break down into its component parts. a. completely b. partly c. never in water, a weak base will break down into its component parts. a. completely b. partly c. never
Answers: 2

Chemistry, 22.06.2019 15:20
An alloy contains 66 g of pure zinc. what is the percentage of zinc in the alloy? express your answer to two significant figures and include the appropriate units.
Answers: 1

Chemistry, 22.06.2019 18:30
How many moles of bromine are needed to produce 3.23 moles of potassium bromide
Answers: 1
You know the right answer?
Copper metal (cu) reacts with silver nitrate (agno3) in aqueous solution to form ag and cu(no3)2. an...
Questions




Mathematics, 03.02.2021 02:50


Mathematics, 03.02.2021 02:50



Computers and Technology, 03.02.2021 02:50


Mathematics, 03.02.2021 02:50



History, 03.02.2021 02:50


Mathematics, 03.02.2021 02:50


Biology, 03.02.2021 02:50


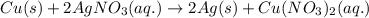
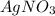 is present in excess, therefore Copper metal is considered as the limiting reagent because it limits the formation of product.
is present in excess, therefore Copper metal is considered as the limiting reagent because it limits the formation of product.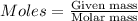 ....(1)
....(1)