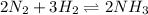Chemistry, 25.07.2019 13:30 pamelperezz26
For the reaction n2(g) + 3h2(g) 2nh3(g), what will happen if more ammonia gas is added? a.)there will be a shift toward the products. b.)the value for keq will change. c.)there will not be a shift in equilibrium. d.)there will be a shift toward the reactants.

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 17:30
The following reaction shows the products when sulfuric acid and aluminum hydroxide react. al(oh)3 + h2so4 → al2(so4)3 + h2o the table shows the calculated amounts of reactants and products when the reaction was conducted in a laboratory. initial mass and yield sulfuric acid aluminum hydroxide initial amount of reactant 40 g 15 g theoretical yield of water from reactant 14.69 g 10.38 g what is the approximate amount of the leftover reactant? 11.73 g of sulfuric acid 10.33 g of sulfuric acid 11.12 g of aluminum hydroxide 13.67 g of aluminum hydroxide
Answers: 1

Chemistry, 22.06.2019 07:20
2pos suppose an object in free fall is dropped from a building. its starting velocity is 0 m/s. ignoring the effects of air resistance, what is the speed (in m/s) of the object after falling 3 seconds? give your answer as a positive decimal without units. answer here
Answers: 1

Chemistry, 22.06.2019 12:30
Acontrol during an experiment. might change remains constant does not exist does change
Answers: 1

You know the right answer?
For the reaction n2(g) + 3h2(g) 2nh3(g), what will happen if more ammonia gas is added? a.)there w...
Questions


Computers and Technology, 03.09.2019 23:30


Computers and Technology, 03.09.2019 23:30


Computers and Technology, 03.09.2019 23:30








Computers and Technology, 03.09.2019 23:30


Computers and Technology, 03.09.2019 23:30