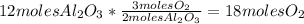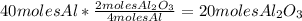Chemistry, 25.07.2019 01:00 ethanhose05
Balance this reaction: al(s) + o2(g) → al2o3(s) how many moles of oxygen will be needed to react with aluminum metal to produce 12.0 moles of aluminum oxide? 6. using the reaction listed in question 5 (above), how many moles of aluminum oxide will be produced by 40.0 moles of aluminum reacting completely with a boat load (that is a lot) of oxygen?

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 23:00
Plz choose one of the compounds from the table and explain how you know the numbers of atoms in your formula. is it possible for two different compounds to be made from the exact same two elements? why or why not? with a limited number of elements (less than 120 are known), does this mean we also have a small number of compounds or do we have a large number of compounds in this world?
Answers: 1

Chemistry, 22.06.2019 06:30
The following reaction shows sodium carbonate reacting with calcium hydroxide. na2co3 + ca(oh)2 → naoh + caco3 how many grams of naoh are produced from 20.0 grams of na2co3? (molar mass of na = 22.989 g/mol, c = 12.01 g/mol, o = 15.999 g/mol, ca = 40.078 g/mol, h = 1.008 g/mol) 12.2 grams 15.1 grams 24.4 grams 30.2 grams
Answers: 2

Chemistry, 22.06.2019 09:00
George is a dalmatian puppy. describe what happens to light that allows you to see george’s black and white coat.
Answers: 1

Chemistry, 22.06.2019 20:30
We are hoping to create 5.72 grams of glucose. the plant was given 4.75 liters of co2 and 2.81 g of h20. which reactant was the limiting reagent? how much excess mass did we have of the other reactant?
Answers: 3
You know the right answer?
Balance this reaction: al(s) + o2(g) → al2o3(s) how many moles of oxygen will be needed to react w...
Questions



Social Studies, 27.01.2020 09:31



Mathematics, 27.01.2020 09:31



Mathematics, 27.01.2020 09:31



History, 27.01.2020 09:31



Computers and Technology, 27.01.2020 09:31

History, 27.01.2020 09:31

Biology, 27.01.2020 09:31

Biology, 27.01.2020 09:31

English, 27.01.2020 09:31

Mathematics, 27.01.2020 09:31