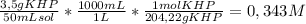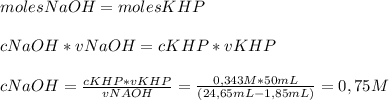To determine the molarity of naoh solution, student took 3.5 g of khp (khp – potassium hydrogen phthalate; molar mass = 204.22 g/mol) and dissolved in 50 ml of water and titrated with the given unknown molarity naoh solution loaded in burette. his burette volume read 1.85 ml at the start of the experiment and 24.65 ml when the phenolphthalein indicator turned pink. using above experimental details, determine the molarity of naoh solution? (enter your answer in two decimal palaces).

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 09:30
Why do cells appear different in distilled water than they do in 10% salt water?
Answers: 2

Chemistry, 22.06.2019 23:10
Amines are good nucleophiles, even though they are neutral molecules. how would the rate of an sn2 reaction between an amine and an alkyl halide be affected if the polarity of the solvent is increased? amines are good nucleophiles, even though they are neutral molecules. how would the rate of an reaction between an amine and an alkyl halide be affected if the polarity of the solvent is increased? because both reactants in the rate-limiting step are neutral, the reaction will be faster if the polarity of the solvent is increased. because both reactants in the rate-limiting step are neutral, the reaction will be slower if the polarity of the solvent is increased. because both reactants in the rate-limiting step are neutral, the reaction will occur at the same rate if the polarity of the solvent is increased. request answer
Answers: 3


You know the right answer?
To determine the molarity of naoh solution, student took 3.5 g of khp (khp – potassium hydrogen phth...
Questions







Mathematics, 24.11.2021 14:00

Physics, 24.11.2021 14:00

English, 24.11.2021 14:00



Social Studies, 24.11.2021 14:00



Mathematics, 24.11.2021 14:00

Advanced Placement (AP), 24.11.2021 14:00


English, 24.11.2021 14:00

Mathematics, 24.11.2021 14:00

Mathematics, 24.11.2021 14:00