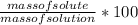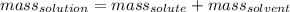Chemistry, 23.07.2019 11:00 vett072804
What is the mass of water required to prepare 250.0 g of 10.0% copper(ii) sulfate solution?

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 15:30
Which of the following mining methods disrupts the sea floor?
Answers: 1

Chemistry, 22.06.2019 05:00
As you watch a surfer ride a wave towards the shoreline, what is the shoreline? a) displacement reference b) reference point c) coordinate plane d) cartesian boundary
Answers: 1

Chemistry, 22.06.2019 09:40
Sulfur dioxide and oxygen react to form sulfur trioxide during one of the key steps in sulfuric acid synthesis. an industrial chemist studying this reaction fills a 25.0l tank with 4.5 mol of sulfur dioxide gas and 4.5 mol of oxygen gas at 30.°c. he then raises the temperature, and when the mixture has come to equilibrium measures the amount of sulfur trioxide gas to be 1.4 mol. calculate the concentration equilibrium constant for the reaction of sulfur dioxide and oxygen at the final temperature of the mixture. round your answer to 2 significant digits.
Answers: 3

Chemistry, 22.06.2019 14:30
In water, a strong acid will break down into its component parts. a. completely b. partly c. never in water, a weak base will break down into its component parts. a. completely b. partly c. never
Answers: 2
You know the right answer?
What is the mass of water required to prepare 250.0 g of 10.0% copper(ii) sulfate solution?...
Questions





Mathematics, 16.09.2019 21:00

Mathematics, 16.09.2019 21:00


Mathematics, 16.09.2019 21:00


History, 16.09.2019 21:00

Biology, 16.09.2019 21:00

Biology, 16.09.2019 21:00

Physics, 16.09.2019 21:00

Chemistry, 16.09.2019 21:00



Social Studies, 16.09.2019 21:00

History, 16.09.2019 21:00

Advanced Placement (AP), 16.09.2019 21:00

Biology, 16.09.2019 21:00