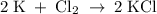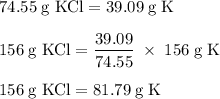Chemistry, 23.07.2019 08:00 treaustin656
What mass of potassium would react with excess chlorine to produce 156 grams of potassium chloride

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 05:40
Calculate: select the worksheet tab. this tab you calculate the analyte concentration. fill in the first set of boxes ("moles h2so4" and "moles naoh") based on the coefficients in the balanced equation. (if there is no coefficient, the value is 1.) record the appropriate volumes in the "ml naoh" and "ml h2so4" boxes. record the concentration of the titrant in the m naoh box. click calculate. what is the concentration listed
Answers: 2

Chemistry, 22.06.2019 10:00
Suppose the universe were completely empty except for one object-a solid sphere moving through space of 100 km/s. what sort of path would the object be moving in? explain your answer
Answers: 1

Chemistry, 22.06.2019 16:00
Answer asap : ( a. how does mucus prevent the entry of pathogens? b. describe two ways white blood cells protect us from pathogens.
Answers: 1

Chemistry, 22.06.2019 21:30
Isopropyl alcohol, (ch3)2choh, is a common solvent. determine the percent by mass of hydrogen in isopropyl alcohol. a) 6.71% h b) 13.4% h c) 25.0% h d) 53.3% h
Answers: 1
You know the right answer?
What mass of potassium would react with excess chlorine to produce 156 grams of potassium chloride...
Questions



Mathematics, 23.01.2020 00:31



Mathematics, 23.01.2020 00:31




Mathematics, 23.01.2020 00:31

Mathematics, 23.01.2020 00:31

Mathematics, 23.01.2020 00:31

Mathematics, 23.01.2020 00:31

Mathematics, 23.01.2020 00:31

Mathematics, 23.01.2020 00:31





English, 23.01.2020 00:31