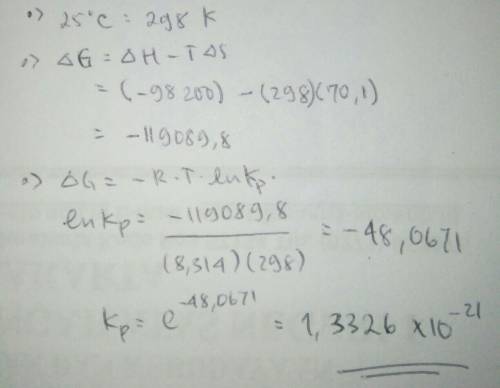Chemistry, 23.07.2019 04:00 kdndiamond9101
Hydrogen peroxide (h2o2) decomposes according to the equation: h2o2(l) ⇆ h2o(l) + ½ o2(g)calculate kp for this reaction at 25°c. (δh° = –98.2 kj/mol, δs° = 70.1 j/k·mol)

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 03:50
Express the following number in scientific notation. 0.026890 =
Answers: 1

Chemistry, 22.06.2019 10:50
How many liters of oxygen gas, at standard temperature and pressure, will react with 35.8 grams of iron metal? 4 fe (s) + 3 o₂ (g) → 2 fe₂o₃ (s)
Answers: 2


Chemistry, 22.06.2019 15:30
The identities of substances are the same before and after which type of change
Answers: 1
You know the right answer?
Hydrogen peroxide (h2o2) decomposes according to the equation: h2o2(l) ⇆ h2o(l) + ½ o2(g)calculate k...
Questions



History, 24.05.2021 14:50



Social Studies, 24.05.2021 14:50

Mathematics, 24.05.2021 14:50


History, 24.05.2021 14:50


English, 24.05.2021 14:50

Health, 24.05.2021 14:50



Mathematics, 24.05.2021 14:50



Mathematics, 24.05.2021 14:50

Chemistry, 24.05.2021 14:50

History, 24.05.2021 14:50