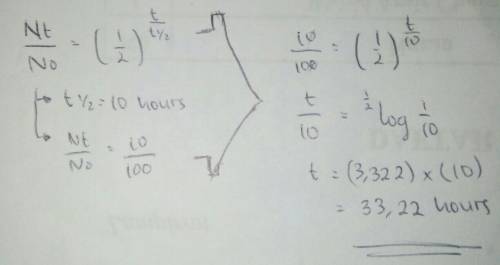Chemistry, 23.07.2019 04:00 pattydixon6
Hydrogen peroxide, which decomposes in a first order reaction, has a half-life of 10 hours in air. how long will it take for hydrogen peroxide to decompose to 10% of its original concentration?

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 00:00
Several kinds of bears are found on earth. most bears are brown or black, but one type of bear, the polar bear, is white. what process led to this difference in fur color? explain your answer.
Answers: 1

Chemistry, 22.06.2019 02:50
The conventional equilibrium constant expression (kc) for the system below is: 2icl(s) ⇄ i2(s) + cl2(g) [cl2] ([i2] + [cl2])/2[icl] [i2][cl2]/[icl]2 none of the listed answers are correct [i2][cl2]/2[icl]
Answers: 2

Chemistry, 22.06.2019 12:00
From the options provided for each element below, choose the properties that it may have based on its location in the periodic table fluorine (f): highly reactive nonmetal shiny a conductor
Answers: 1

Chemistry, 22.06.2019 20:00
Many free radicals combine to form molecules that do not contain any unpaired electrons. the driving force for the radical–radical combination reaction is the formation of a new electron‑pair bond. consider the chemical equation. n(g)+no(g)⟶nno(g) n(g)+no(g)⟶nno(g) write lewis formulas for the reactant and product species in the chemical equation. include nonbonding electrons. n(g)n(g) select draw rings more erase select draw rings more erase select draw rings more erase n no(g)
Answers: 1
You know the right answer?
Hydrogen peroxide, which decomposes in a first order reaction, has a half-life of 10 hours in air. h...
Questions

Social Studies, 07.07.2019 01:00



Geography, 07.07.2019 01:00

Mathematics, 07.07.2019 01:00



Social Studies, 07.07.2019 01:00

Mathematics, 07.07.2019 01:00



Biology, 07.07.2019 01:00


Physics, 07.07.2019 01:00


Health, 07.07.2019 01:00

Computers and Technology, 07.07.2019 01:00

Mathematics, 07.07.2019 01:00


English, 07.07.2019 01:00