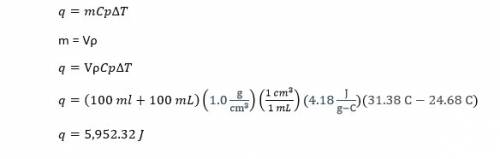In a coffee-cup calorimeter, 100.0 ml of 1.0 m naoh and 100.0 ml of 1.0 m hcl are mixed. both solutions were originally at 24.68c. after the reaction, the final temperature is 31.38c. assuming that all the solutions have a density of 1.0 g/cm3 and a specific heat capacity of 4.18 j/8c ? g, calculate the enthalpy change for the neutralization of hcl by naoh

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 20:00
Drag each number to the correct location on the equation. each number can be used more than once, but not all numbers will be used. balance the equation with the coefficients. 2 3 4 5 kclo3 -> kcl + o2
Answers: 1

Chemistry, 22.06.2019 09:50
Although respiratory organs vary across different organisms, they all contain respiratory surfaces that have a large surface area and are extremely thin. explain why having an extremely thin respiratory surface with a large surface area is advantageous for the process of gas exchange
Answers: 1

Chemistry, 22.06.2019 13:00
These questions are based on the attached photo. the experiment is about burning magnesium metal with oxygen. 1. write the balanced chemical equation for the reaction you are performing. 2. calculate the mass of magnesium metal used in each trial. o trial 1: o trial 2: 3. calculate the actual yield of magnesium oxide for each trial. o trial 1: o trial 2: 4. magnesium is the limiting reactant in this experiment. calculate the theoretical yield of mgo for each trial. o trial 1: o trial 2: 5. determine the percent yield of mgo for your experiment for each trial. o trial 1: o trial 2: 6. determine the average percent yield of mgo for the two trials. your company currently uses a process with a similar cost of materials that has an average percent yield of 91 percent. if the average percent yield of this process is higher than that, this could save the company money. what is your recommendation to the company? support your recommendation using your data, calculations, and understanding of stoichiometry gathered from this lab.
Answers: 1

You know the right answer?
In a coffee-cup calorimeter, 100.0 ml of 1.0 m naoh and 100.0 ml of 1.0 m hcl are mixed. both soluti...
Questions


History, 17.12.2020 18:50



Social Studies, 17.12.2020 18:50

Biology, 17.12.2020 18:50

Advanced Placement (AP), 17.12.2020 18:50




Mathematics, 17.12.2020 18:50


Mathematics, 17.12.2020 18:50


English, 17.12.2020 18:50

Arts, 17.12.2020 18:50


Computers and Technology, 17.12.2020 18:50

Mathematics, 17.12.2020 18:50

English, 17.12.2020 18:50