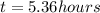How long must a constant current of 50.0 a be passed through an electrolytic cell containing aqueous cu2+ ions to produce 5.00 moles of copper metal? how long must a constant current of 50.0 a be passed through an electrolytic cell containing aqueous cu2+ ions to produce 5.00 moles of copper metal? 5.36 hours 2.68 hours 0.373 hours 0.187 hours?

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 06:30
Predict whether the changes in enthalpy, entropy, and free energy will be positive or negative for the boiling of water, and explain your predictions. how does temperature affect the spontaneity of this process?
Answers: 1

Chemistry, 22.06.2019 13:20
Can someone me with 3 and 4 plz. this is for masteries test.
Answers: 2

Chemistry, 22.06.2019 20:00
Many free radicals combine to form molecules that do not contain any unpaired electrons. the driving force for the radical–radical combination reaction is the formation of a new electron‑pair bond. consider the chemical equation. n(g)+no(g)⟶nno(g) n(g)+no(g)⟶nno(g) write lewis formulas for the reactant and product species in the chemical equation. include nonbonding electrons. n(g)n(g) select draw rings more erase select draw rings more erase select draw rings more erase n no(g)
Answers: 1

You know the right answer?
How long must a constant current of 50.0 a be passed through an electrolytic cell containing aqueous...
Questions






English, 10.03.2020 03:43


Health, 10.03.2020 03:43






Mathematics, 10.03.2020 03:44

English, 10.03.2020 03:44





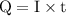
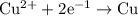
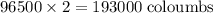
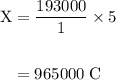
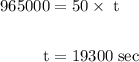
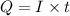
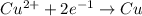
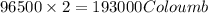 of electricity deposits 1 mole of Cu.
of electricity deposits 1 mole of Cu.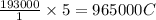
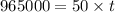
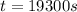 (1 hour=3600sec)
(1 hour=3600sec)