
Chemistry, 21.07.2019 23:00 dajeourcrazy15
A5.018 gram sample of a certain hydrate of magnesium sulfate, mgso4•xh2o, is heated until all the water is driven off. the resulting anhydrous compound weighs 2.449 grams. what is the formula of the hydrate?

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 05:00
Given sno2 + 2h2 - sn + 2h20 tin oxide reacts with hydrogen to produce tin and water. how many moles of sno2 are needed to produce 500.0 grams of sn?
Answers: 3


Chemistry, 22.06.2019 18:00
How does climate change cause the ocean's thermohaline current to slow down?
Answers: 3

Chemistry, 22.06.2019 20:00
In vapor-liquid equilibrium in a binary mixture, both components are generally present in both phases. how many degrees of freedom are there for such a system? the reaction between nitrogen and hydrogen to form ammonia occurs in the gas phase. how many degrees of freedom are there for this system? steam and coal react at high temperatures to form hydrogen, carbon monoxide, carbon dioxide, and methane. the following reactions have been suggested as being involved in the chemical transformation:
Answers: 3
You know the right answer?
A5.018 gram sample of a certain hydrate of magnesium sulfate, mgso4•xh2o, is heated until all the wa...
Questions





Chemistry, 26.01.2020 23:31

History, 26.01.2020 23:31







Physics, 26.01.2020 23:31


Mathematics, 26.01.2020 23:31

English, 26.01.2020 23:31



Spanish, 26.01.2020 23:31

English, 26.01.2020 23:31

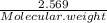
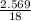
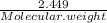
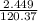
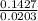 = 7.02 ~7
= 7.02 ~7

