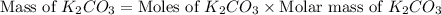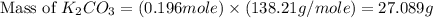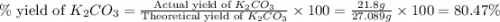Chemistry, 21.07.2019 23:00 asuhdude57
)determine the theoretical yield and the percent yield if 21.8 g of k2co3 is produced from reacting 27.9 g ko2 with 29.0 l of co2 (at stp). the molar mass of ko2 = 71.10 g/mol and k2co3 = 138.21 g/mol.

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 15:00
Which element in the third period would you expect to have the larger atomic radius, sodium (na) or sulfur (s)? a. sodium, because it has a higher effective nuclear charge attracting electrons in fewer energy levels. b. sodium, because it has fewer protons attracting electrons in the same energy levels. c. sulfur, because it has more protons attracting electrons in more energy levels. d. sulfur, because it has a higher effective nuclear charge attracting electrons in the same energy levels.
Answers: 2



Chemistry, 22.06.2019 10:30
Great amounts of electromagnetic energy from our sun and other bodies in space travel through space. which is a logical conclusion about these electromagnetic waves? their energy must be very their frequency must be very low these waves can travel without a medium they only travel through a vacuum of space
Answers: 2
You know the right answer?
)determine the theoretical yield and the percent yield if 21.8 g of k2co3 is produced from reacting...
Questions


Social Studies, 05.05.2021 16:00



Mathematics, 05.05.2021 16:00


Mathematics, 05.05.2021 16:00


Chemistry, 05.05.2021 16:00

Physics, 05.05.2021 16:00

Mathematics, 05.05.2021 16:00

Mathematics, 05.05.2021 16:00

Chemistry, 05.05.2021 16:00

History, 05.05.2021 16:00



Arts, 05.05.2021 16:00

English, 05.05.2021 16:00

Mathematics, 05.05.2021 16:00

Chemistry, 05.05.2021 16:00

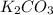 = 27.089 g
= 27.089 g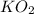 = 27.9 g
= 27.9 g
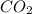 = 29.0 L (At STP)
= 29.0 L (At STP)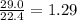 mole of
mole of 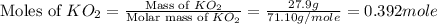
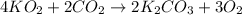
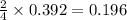 moles of
moles of