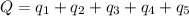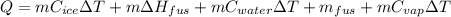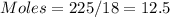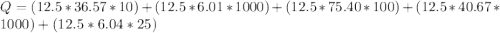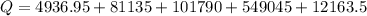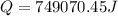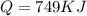Engineering, 07.11.2019 22:31 nano792001
Calculate the amount of energy in kilojoules needed to change 225 g of water ice at −10 ∘c to steam at 125 ∘c. the following constants may be useful: cm (ice)=36.57 j/(mol⋅∘c) cm (water)=75.40 j/(mol⋅∘c) cm (steam)=36.04 j/(mol⋅∘c) δhfus=+6.01 kj/mol δhvap=+40.67 kj/mol express your answer with the appropriate units.

Answers: 2


Another question on Engineering

Engineering, 04.07.2019 18:10
What are the two (02) benefits, which may result from a successful implementation of preventive maintenance (pm) program in an organization? (clo3)a)- lean manufacturing b)-overlapping responsibilities c)-the planner is not qualified d)-accurate contractor information e)-reduction in equipment redundancies f)-accurate stores information
Answers: 3

Engineering, 04.07.2019 19:10
Acircular aluminum shaft mounted in a journal is shown. the symmetric clearance gap between the shaft and journal is filled with sae 10w-30 oil at t 30°c. the shaft is caused to turn by the attached mass and cord. develop and solve a differential equation for the angular speed of the shaft as a function of time.
Answers: 2

Engineering, 04.07.2019 19:20
Heat transfer by is the fastest mode of heat transfer that requires no intervening medium. a)-conduction b)-convection c)-radiation d)-conduction and convection
Answers: 1

Engineering, 04.07.2019 19:20
Determine the stoichiometric and actual air-fuel ratios and the mole flue gas composition for combustion with 15% excess air for raw indiana, pennsylvania raw coal.
Answers: 3
You know the right answer?
Calculate the amount of energy in kilojoules needed to change 225 g of water ice at −10 ∘c to steam...
Questions


Mathematics, 23.11.2021 04:50


History, 23.11.2021 04:50





Social Studies, 23.11.2021 04:50

Mathematics, 23.11.2021 04:50


Computers and Technology, 23.11.2021 04:50

Mathematics, 23.11.2021 04:50

Mathematics, 23.11.2021 04:50


History, 23.11.2021 04:50

English, 23.11.2021 04:50

Spanish, 23.11.2021 04:50

Mathematics, 23.11.2021 04:50