
Engineering, 27.03.2020 21:29 connermichaela
A quantity of nitrogen gas in a piston–cylinder assembly undergoes a process at a constant pressureof 80 bar from 220 to 300 K. Determine the work and heat transfer for the process, each in kJ per kmol of nitrogen.

Answers: 3


Another question on Engineering

Engineering, 04.07.2019 18:10
Carbon dioxide gas expands isotherm a turbine from 1 mpa, 500 k at 200 kpa. assuming the ideal gas model and neglecting the kinetic and potential energies, determine the change in entropy, heat transfer and work for each kilogram of co2.
Answers: 2

Engineering, 04.07.2019 18:10
Courses that are developed by subject matter experts, internal or extemal to the college or university. these programs are marketed by the school (clo2) marks a)-vocational schools b)-vendor training c)-colleges & universities d)-continuing education programs
Answers: 2

Engineering, 04.07.2019 18:20
An engine runs on the ideal diesel cycle. the cycle has a compression ratio of 20 and a cutoff ratio of 2. the highest temperature in the cycle is 1200 k. if the heat into the system is 300 kj/kg of working fluid and using variable specific heats determine the work produced per mass of working fluid
Answers: 3

Engineering, 04.07.2019 18:20
How much power could a wind turbine produce if it had the following specifications? cp = 0.45 -d=1.2kg/m3 d=50m v 5m/s
Answers: 2
You know the right answer?
A quantity of nitrogen gas in a piston–cylinder assembly undergoes a process at a constant pressureo...
Questions

History, 21.07.2019 02:00

History, 21.07.2019 02:00







English, 21.07.2019 02:00


History, 21.07.2019 02:00

World Languages, 21.07.2019 02:00

Geography, 21.07.2019 02:00


Computers and Technology, 21.07.2019 02:00

History, 21.07.2019 02:00


Biology, 21.07.2019 02:00


Physics, 21.07.2019 02:00

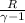 × (t2-t1) + 665.12
× (t2-t1) + 665.12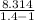 × (300-220) + 665.12
× (300-220) + 665.12 

