
Engineering, 18.02.2021 07:10 mrus
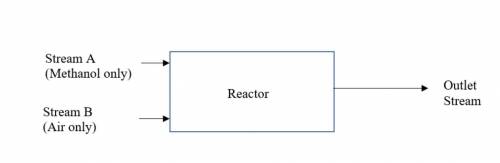
A petrochemical plant in Island C produces formaldehyde (CH2O) industrially by the
catalytic oxidation of methanol (CH3OH). The following is the chemical reaction:
CH3OH + ½O2 → CH2O + H2O --- (1)
Unfortunately, a significant portion of the formaldehyde will react with oxygen to produce CO
and H2O. The following is the side reaction:
CH2O + ½O2 → CO + H2O --- (2)
Assume twice the stoichiometric amount of air needed for oxidation (all chemical reactions) is
fed to the reactor. The conversion of methanol is 90% in the reactor. The outlet stream of the
reactor was analysed and found to be 150 kg/hr of formaldehyde, 30 kg/hr of CO and other
components.(i) Determine the mass flowrate (kg/hr) of Stream A. (ii) Determine the molar flowrate (kg-moles/hr) of Stream B (iii) Determine the mass flowrate (kg/hr) of water in the outlet stream.

Answers: 3


Another question on Engineering

Engineering, 04.07.2019 18:10
Thermal stresses are developed in a metal when its a) initial temperature is changed b) final temperature is changed c) density is changed d) thermal deformation is prevented e) expansion is prevented f) contraction is prevented
Answers: 2

Engineering, 04.07.2019 18:10
Slip occurs via two partial dislocations because of (a) the shorter path of the partial dislocation lines; (b) the lower energy state through partial dislocations; (c) the charge balance.
Answers: 1

Engineering, 04.07.2019 18:10
Condition monitoring is a major component of. (clo4) a)- predictive maintenance. b)-preventive maintenance c)-proactive maintenance d)-reactive maintenance.
Answers: 1

Engineering, 04.07.2019 19:10
The proportional limit is always greater than the yield strength for a material. a)-trune b)- false
Answers: 3
You know the right answer?
A petrochemical plant in Island C produces formaldehyde (CH2O) industrially by the
catalytic oxidat...
Questions


Mathematics, 19.04.2021 16:30





English, 19.04.2021 16:30

Biology, 19.04.2021 16:30





Mathematics, 19.04.2021 16:30

Mathematics, 19.04.2021 16:30



Advanced Placement (AP), 19.04.2021 16:30

Mathematics, 19.04.2021 16:30




