Part a) 2,000 mL of solution A
Part b) 40 mL of solution A and 60 mL of solution B
Part c) Is not possible
Step-by-step explanation:
Part a) How many mL of Solution A must be added to 500 mL of Solution B in order to produce a 70% acid solution?
Remember that
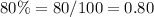
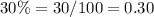
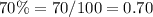
Let
x ---->mL of solution A n 80% acid solution
y ---->mL of solution B n 30% acid solution
we know that
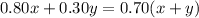
Remember that
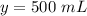 ----> equation B
----> equation B
substitute equation B in equation A

solve for x

Part b) How many mL of Solution A and how many mL of Solution B must be combined to form a 100 mL solution that is 50% acid?
Remember that
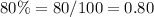
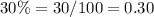
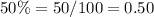
Let
x ---->mL of solution A n 80% acid solution
y ---->mL of solution B n 30% acid solution
we know that
 ----> equation A
----> equation A
 -----> equation B
-----> equation B
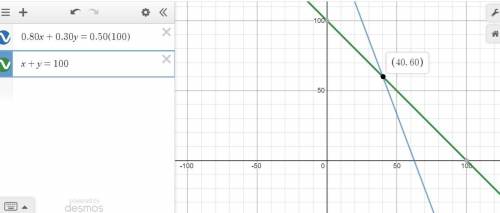
Solve the system by graphing
Remember that the solution is the intersection point both graphs
using a graphing tool
The solution is (40,60)
see the attached figure
therefore
40 mL of solution A and 60 mL of solution B
Part c) Is there a combination of Solution A and Solution B that is 90% acid?
Is not possible , because 90% is greater than 80% of solution A and greater than 30% of solution B
The percentage of the final concentration must be less than 80% and more than 30%