Chemical reaction:
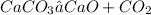
Molar mass of;CaCO3 = 100gCaO = 56gCO2= 44gAccording to law of conservation of mass:
Mass of CaCO3 = Mass of CaO+Mass of CO2
56g of CaO is obtained from decomposition of 100g of CaCO3.
So,
1.4g of CaO is obtained from decomposition of ↴

Mass of CO2

Therefore, The mass of CaO is 2.5g and mass of CO2 is 1.1g respectively!

 Q3
Q3


























