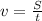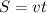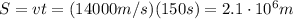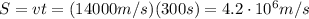Answers: 1


Another question on Physics

Physics, 22.06.2019 06:20
Clothing made of several thin layers of fabric with trapped air in between, often called ski clothing, is commonly used in cold climates because it is light, fashionable, and a very effective thermal insulator. so it is no surprise that such clothing has largely replaced thickand heavy old-fashioned coats. (a) consider a jacket made of five layers of 0.1-mm-thick synthetic fabric (k = 0.13 w/m·°c) with 1.5-mm-thick air space (k = 0.026 w/m·°c) between the layers. assuming the inner surface temperature of the jacket to be 28°c and the surface area to be 1.25 m2, determine the rate of heat loss through the jacket when the temperature of the outdoors is 0°c and the heat transfer coefficient at the outer surface is 25 w/m2·°c. (b) what would your response be if the jacket is made of a single layer of 0.5-mm-thick synthetic fabric? what should be the thickness of a wool fabric (k = 0.035 w/m·°c) if the person is to achieve the same level of thermal comfort wearing a thick wool coat instead of a five-layer ski jacket?
Answers: 1

Physics, 22.06.2019 15:00
Amoving company collects data about couches they move and displays the data in a table. the force along the ramp, the angle of the ramp, and the height the couch is raised to are shown. which statements correctly compare the work done to move the couches? check all that apply. the same amount of work is done for couches 1 and 3. the same amount of work is done for couches 2 and 3. the work for couch 1 is greater than the work for couch 2. the work for couch 2 is less than the work for couch 3. the work for couch 3 is greater than the work for couch 1. the work for couch 2 is greater than the work for couch 1.
Answers: 1

Physics, 22.06.2019 17:00
Since the energy was not raising the temperature of the water during these segments, what was the energy used to do instead? your answer must include an explanation of what is happening to the molecules.
Answers: 3

Physics, 23.06.2019 08:30
[05.02] what is the best explanation of the kinetic molecular theory as it relates to the energy of the molecules in the states of matter? (1 point) the molecules in a gas have greater kinetic energy than those in a liquid, which in turn have greater kinetic energy than the molecules in a solid. kinetic energy cannot be created nor destroyed, and, as a result, all states of matter have the same kinetic energy. the molecules in a gas have greater kinetic energy; therefore, they are more tightly packed than the molecules in either a liquid or a solid. the molecules in gases and liquids, in constant motion, have greater kinetic energy than those in a solid, which do not move or vibrate because their position is fixed.
Answers: 2
You know the right answer?
Arocket travels at a speed of 14,000 m/s. what is the distance travelled by the rocket in 150s? wha...
Questions





Mathematics, 20.09.2019 11:30

Mathematics, 20.09.2019 11:30


Biology, 20.09.2019 11:30

Chemistry, 20.09.2019 11:30

Computers and Technology, 20.09.2019 11:30

Mathematics, 20.09.2019 11:30


Mathematics, 20.09.2019 11:30


Mathematics, 20.09.2019 11:30

Mathematics, 20.09.2019 11:30


Mathematics, 20.09.2019 11:30

Biology, 20.09.2019 11:30

Chemistry, 20.09.2019 11:30