
Calculate the energy in the form of heat (in kJ) required to change 75.0 g of liquid water at 27.0 °C to ice at –20.0 °C. Assume that no energy in the form of heat is transferred to the environment. (Heat of fusion = 333 J/g; heat of vaporization = 2256 J/g; specific heat capacities: ice = 2.06 J/g·K, liquid water = 4.184 J/g·K)

Answers: 3


Another question on Physics

Physics, 22.06.2019 17:00
Two manned satellites approaching one another at a relative speed of 0.550 m/s intend to dock. the first has a mass of 2.50 ✕ 103 kg, and the second a mass of 7.50 ✕ 103 kg. assume that the positive direction is directed from the second satellite towards the first satellite. (a) calculate the final velocity after docking, in the frame of reference in which the first satellite was originally at rest.(b) what is the loss of kinetic energy in this inelastic collision? (c) repeat both parts, in the frame of reference in which the second satellite was originally at rest. final velocity(d) loss of kinetic energy = ?
Answers: 2

Physics, 23.06.2019 00:10
An astronaut floating in space is trying to use her jetpack to get back to her space station, but she is being pulled away by a nearby planet, as shown in the image below. her jetpack provides a constant thrust of 310 n. if she angles her jetpack in such a way that it cancels out the vertical force due to the planet's gravity, what is her net horizontal force? thaud 487 o a. 54.8 n toward the space station o b. 212.9 n toward the space station o c. 185.2 n toward the space station o d 956 n toward the space station
Answers: 1


Physics, 23.06.2019 03:30
Ineed to answer this problem but i lack in knowledge, what should i study to answer this problem? i have never used derivatives in a physics problem so i'm not sure what's happening and i can't find videos explaining.
Answers: 2
You know the right answer?
Calculate the energy in the form of heat (in kJ) required to change 75.0 g of liquid water at 27.0 °...
Questions









History, 10.03.2020 19:22



Law, 10.03.2020 19:22

English, 10.03.2020 19:22

Mathematics, 10.03.2020 19:22



Computers and Technology, 10.03.2020 19:22


Biology, 10.03.2020 19:22

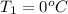
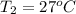
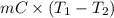
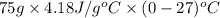
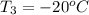 = (20 + 273) K = 293 K and specific heat of ice is 2.108 kJ/kg K.
= (20 + 273) K = 293 K and specific heat of ice is 2.108 kJ/kg K. 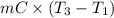
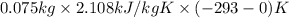
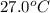 to ice at
to ice at 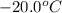 is -37.86 kJ.
is -37.86 kJ.

