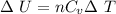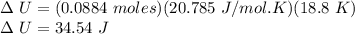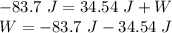0.0884 moles of a diatomic gas
are in a piston. When the piston
is compressed, the temperatur...

Physics, 12.12.2020 17:00 ineedtopeebeforethec
0.0884 moles of a diatomic gas
are in a piston. When the piston
is compressed, the temperature
drops by 18.8 K, and 83.7 J of
heat flow out. Find W.
(Be careful with + and - signs.
+W = expansion, +Q = added,
+AU = temp goes up)
(Unit = J)

Answers: 2


Another question on Physics


Physics, 22.06.2019 15:20
Abag of potato chips contains 2.00 l of air when it is sealed at sea level at a pressure of 1.00 atm and a temperature of 20.0 deg c. what will be the volume of the air in the bag if you take it with you, still sealed, to the mountains where the temperature is 7.00 deg c and atmospheric pressure is 70.0 kpa? assume that the bag behaves like a balloon and that the air in the bag is in thermal equilibrium with the outside air.
Answers: 3


Physics, 22.06.2019 20:10
Consider two less-than-desirable options. in the first you are driving 30 mph and crash head-on into an identical car also going 30 mph. in the second option you are driving 30 mph and crash head-on into a stationary brick wall. in neither case does your car bounce off the thing it hits, and the collision time is the same in both cases. which of these two situations would result in the greatest impact force?
Answers: 1
You know the right answer?
Questions



Social Studies, 06.10.2019 21:30

History, 06.10.2019 21:30


Chemistry, 06.10.2019 21:30


Geography, 06.10.2019 21:30

Mathematics, 06.10.2019 21:30

Health, 06.10.2019 21:30


Biology, 06.10.2019 21:30


Mathematics, 06.10.2019 21:30

Mathematics, 06.10.2019 21:30

Health, 06.10.2019 21:30

Mathematics, 06.10.2019 21:30


Biology, 06.10.2019 21:30

English, 06.10.2019 21:30