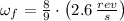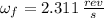Answers: 1


Another question on Physics

Physics, 22.06.2019 00:10
The energy released by a chemical reaction can be measured using a calorimeter. when barium hydroxide octahydrate crystals are reacted with dry ammonium chloride inside of a coffee cup calorimeter, the temperature of the 18.00 g of water in the calorimeter decreases from 30.0°c to 8.0°c. the equation for calculating energy absorbed or released by a reaction is: where q is the energy released or absorbed, m is the mass of water in the calorimeter, cp is the specific heat of water, and δt is the observed temperature change. if the specific heat of liquid water is 4.19 j/g·°c, how much energy was absorbed by the reaction?
Answers: 3

Physics, 22.06.2019 03:30
Calculate the mass of an object that has a momentum of 100kg x m/sec and velocity of 4 m/sec
Answers: 1


Physics, 22.06.2019 18:00
Which is the most accurate name for the ionic compound cas?
Answers: 1
You know the right answer?
A solid disk with a mass of 0.8 kg and a radius of 12cm is rotating at a rate of 2.6 rev/s. Another...
Questions


Mathematics, 07.01.2020 20:31


History, 07.01.2020 20:31






Mathematics, 07.01.2020 20:31

Chemistry, 07.01.2020 20:31




Chemistry, 07.01.2020 20:31

History, 07.01.2020 20:31



English, 07.01.2020 20:31

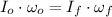 (1)
(1)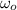 ,
, 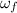 - Initial and final angular speeds, measured in revolutions per second.
- Initial and final angular speeds, measured in revolutions per second.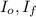 - Initial and final moments of inertia, measured in kilograms per square meter.
- Initial and final moments of inertia, measured in kilograms per square meter.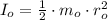 (2)
(2)![I_{f} = \frac{1}{2}\cdot \left[m_{o}\cdot r_{o}^{2}+\frac{1}{2}\cdot m_{o}\cdot \left(\frac{1}{2}\cdot r_{o} \right)^{2}\right]](/tpl/images/0995/3231/47616.png)
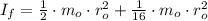
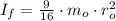 (3)
(3)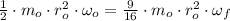
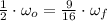
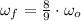 (4)
(4)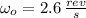 , then the new angular speed of both disks when they are rotating together is:
, then the new angular speed of both disks when they are rotating together is: