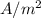Answers: 2


Another question on Physics

Physics, 21.06.2019 17:00
These models show the electron structures of two different nonmetal elements. which element is likely more reactive, and why? element 1 is more reactive because it has fewer electron shells and is toward the top of its group on the periodic table. element 1 is more reactive because it has more electrons in its valence shell and is farther to the right on the periodic table. element 2 is more reactive because it does not have a valence shell close to the nucleus, so it will attract electrons. element 2 is more reactive because it does not have a full valence shell, so it will attract electrons.
Answers: 2



Physics, 22.06.2019 08:30
What properties of a moving object are used in determining the object's energy of motion
Answers: 2
You know the right answer?
Dimensional formula of current density...
Questions


Chemistry, 22.01.2020 08:31





Social Studies, 22.01.2020 08:31


Mathematics, 22.01.2020 08:31


Mathematics, 22.01.2020 08:31

Mathematics, 22.01.2020 08:31


Mathematics, 22.01.2020 08:31

Mathematics, 22.01.2020 08:31

Mathematics, 22.01.2020 08:31


Mathematics, 22.01.2020 08:31

History, 22.01.2020 08:31

Mathematics, 22.01.2020 08:31

![[M ^{-2} A]](/tpl/images/0500/1066/0faff.png)