Answers: 1


Another question on Physics

Physics, 22.06.2019 00:10
The energy released by a chemical reaction can be measured using a calorimeter. when barium hydroxide octahydrate crystals are reacted with dry ammonium chloride inside of a coffee cup calorimeter, the temperature of the 18.00 g of water in the calorimeter decreases from 30.0°c to 8.0°c. the equation for calculating energy absorbed or released by a reaction is: where q is the energy released or absorbed, m is the mass of water in the calorimeter, cp is the specific heat of water, and δt is the observed temperature change. if the specific heat of liquid water is 4.19 j/g·°c, how much energy was absorbed by the reaction?
Answers: 3


Physics, 22.06.2019 09:30
Asap i'm in class rn a 1,000-kg car is traveling 20 m/s on a flat stretch of road. it gets to a hill and coasts uphill until it stops. how high up the hill does the car travel? givens: equation: 1/2mv2initial=mghfinal solve for h. plug & chug, label.
Answers: 2

Physics, 22.06.2019 16:30
In a classical model of the hydrogen atom, the electron moves around the proton in a circular orbit of radius 0.053 nm. what is the electron's orbital frequency? what is the effective current of the electron?
Answers: 3
You know the right answer?
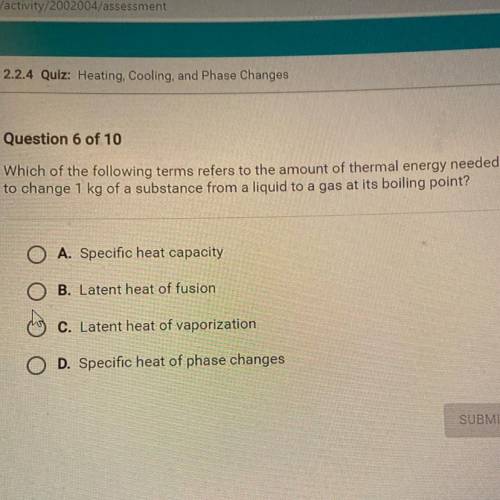
which of the following terms refers to the amount of thermal energy needed to change 1kg of a a subs...
Questions




Biology, 09.12.2020 22:50



Mathematics, 09.12.2020 22:50

Mathematics, 09.12.2020 22:50



Mathematics, 09.12.2020 22:50



Mathematics, 09.12.2020 23:00




Physics, 09.12.2020 23:00

Mathematics, 09.12.2020 23:00