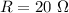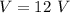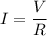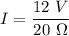Answers: 1


Another question on Physics

Physics, 22.06.2019 00:40
Electroplating is a way to coat a complex metal object with a very thin (and hence inexpensive) layer of a precious metal, such as silver or gold. in essence the metal object is made the cathode of an electrolytic cell in which the precious metal cations are dissolved in aqueous solution. suppose a current of 480.ma is passed through an electroplating cell with an aqueous solution of agno3 in the cathode compartment for 46.0 seconds. calculate the mass of pure silver deposited on a metal object made into the cathode of the cell. round your answer to 3 significant digits. also, be sure your answer contains a unit symbol. ×10μ
Answers: 3

Physics, 22.06.2019 11:30
(2) (a) you have a simple circuit that consists of only a battery (δvbat=1.5v) and two resistors with resistance r1=10ω and r2=5ω, connected in series with each other. what is the current running through the battery? (b) you re-arrange your circuit so now r2 is attached in parallel to r1 rather than in series. what is the current running through the battery? (c) you add an additional resistor r3=7ω on the same branch as r2. what is the current running through the battery? (d) what is the power dissipated in r3?
Answers: 3

Physics, 22.06.2019 12:00
This is for my 8th grade science class, i really don’t understand. can someone me? , lol.
Answers: 1

Physics, 22.06.2019 22:30
Which is a substance that can be separated by physical means and is not the same throughout
Answers: 2
You know the right answer?
Awire with a resistance of 20 ω is connected to a 12-v battery. what is the current flowing through...
Questions

Mathematics, 21.01.2021 02:00

Mathematics, 21.01.2021 02:00

History, 21.01.2021 02:00



History, 21.01.2021 02:00

Mathematics, 21.01.2021 02:00


English, 21.01.2021 02:00



World Languages, 21.01.2021 02:00

Biology, 21.01.2021 02:00


Mathematics, 21.01.2021 02:00

History, 21.01.2021 02:00



Mathematics, 21.01.2021 02:00

Mathematics, 21.01.2021 02:00