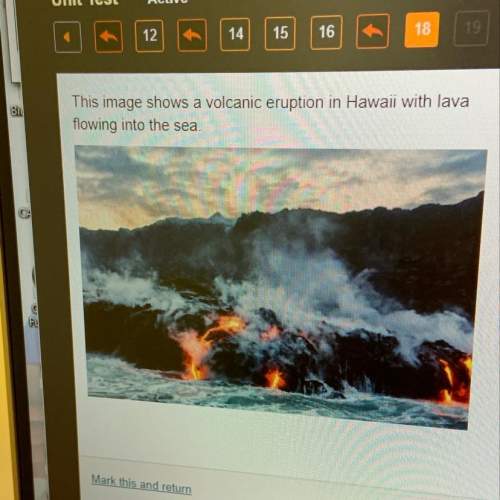
When solid magnesium burns, it reacts with oxygen in the air to form a powder called magnesium oxide. A chemist performed this reaction in a lab and found that the mass of the magnesium oxide was greater than the mass of the magnesium. What is the reason behind this increase in mass? A. The increase in the mass of the magnesium oxide was due to magnesium atoms in the air. B. The increase in the mass of the magnesium oxide was due to oxygen atoms in the air. C. The increase in the mass of the magnesium oxide happened because magnesium is heavier than oxygen. D. The increase in the mass of the magnesium oxide happened because oxygen is heavier than magnesium.

Answers: 2


Another question on Biology

Biology, 22.06.2019 15:00
If you relate the doppler effect on sound to the color of light waves, a police car would be the color as it approaches and the color as it recedes away from the observer.
Answers: 3


Biology, 22.06.2019 19:00
Why does controlling transcription affect prokaryotic gene expression?
Answers: 3

Biology, 23.06.2019 03:00
Achemical substance made by glands in the endocrine system that travel through the bloodstream and affects cells and tissues throughout the body ?
Answers: 1
You know the right answer?
When solid magnesium burns, it reacts with oxygen in the air to form a powder called magnesium oxide...
Questions

English, 04.09.2019 04:20

English, 04.09.2019 04:20





History, 04.09.2019 04:20

Computers and Technology, 04.09.2019 04:20





Chemistry, 04.09.2019 04:30

History, 04.09.2019 04:30

Mathematics, 04.09.2019 04:30




Mathematics, 04.09.2019 04:30

Mathematics, 04.09.2019 04:30