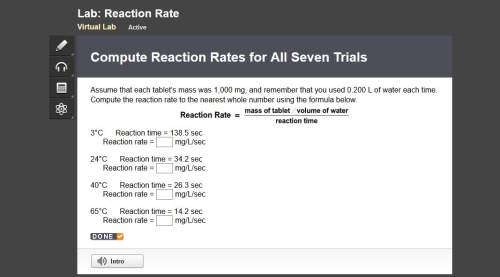
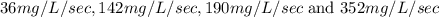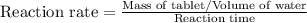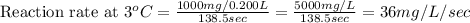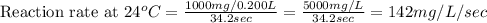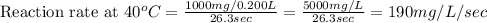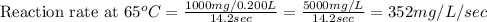Compute reaction rates for all seven trials
reaction rate is usually computed as a change in co...

Chemistry, 27.11.2019 06:31 luvpeaceandsocc3678
Compute reaction rates for all seven trials
reaction rate is usually computed as a change in concentration (e. g., molarity) per change in time. the tablet is not pure nahco3, so the molarity cannot be computed accurately. (in addition, the quantities in this lab are quite small, and the resulting values would be small decimal values.) instead, compute the following as a measurement of reaction rate.

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 02:20
Compared with the freezing-point depression of a 0.01 m c6h12o6 solution, the freezing-point depression of a 0.01 m nacl solution is
Answers: 1

Chemistry, 22.06.2019 03:00
In the 1800s, one of the statements in john dalton's atomic theory was that atoms are indivisible. later experimental evidence led to the discovery of subatomic particles such as neutrons, electrons, and protons. what happened to the indivisible atom part of dalton's atomic theory, and why?
Answers: 3

Chemistry, 22.06.2019 11:00
Iron (3) oxide will decompose in the presence of hydrogen gas and heater to produced iron and digydrogen monoxide white a balanced chemical equation
Answers: 1

Chemistry, 22.06.2019 22:30
[ou.03jthe pictures below show the wavelengths and intensities of electromagnetic radiations emitted by three stars, star 1, star 2, and star 3. intensity intensity- intensity- 1000 3500 6000 8500 11000 wavelength (a) star 1 1000 3500 6000 8500 11000 1000 3500 6000 8500 11000 wavelength (a) wavelength (a) star 2 star 3 which of these statements is correct about the color of the three stars? star 2 is white in color o star 2 is yellow in color star 1 and star 3 are yellow in color star 1 and star 3 are white in color
Answers: 1
You know the right answer?
Questions




Mathematics, 20.09.2019 18:30

Physics, 20.09.2019 18:30

English, 20.09.2019 18:30


Biology, 20.09.2019 18:30

Mathematics, 20.09.2019 18:30

History, 20.09.2019 18:30

Mathematics, 20.09.2019 18:30

Biology, 20.09.2019 18:30




Mathematics, 20.09.2019 18:30




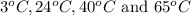 are
are