Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 02:50
Using a value of ksp = 1.8 x 10-2 for the reaction pbcl2 pb+2(aq) + 2cl -(aq). if the value of ksp was determined to be only 1.2 x 10-2: too much solid has dissolved. additional precipitate is forming. the solution is unsaturated. the ions are now combining to reduce their concentrations.
Answers: 3

Chemistry, 22.06.2019 17:00
Which statement is true about a catalyst? a: a catalyst decreases the rate of the reaction. b. a catalyst is consumed during a chemical reaction. c. a catalyst lowers the activation energy of a reaction. d. a catalyst increases the reactant concentration during a reaction.
Answers: 1

Chemistry, 23.06.2019 01:20
How can parts of a solution be separated by chromatography?
Answers: 1

Chemistry, 23.06.2019 01:30
At a certain temperature the rate of this reaction is first order in hi with a rate constant of : 0.0632s2hig=h2g+i2g suppose a vessel contains hi at a concentration of 1.28m . calculate how long it takes for the concentration of hi to decrease to 17.0% of its initial value. you may assume no other reaction is important. round your answer to 2 significant digits.
Answers: 1
You know the right answer?
Use the e0 values of the following elements: zn, cu, pb, and al.
copper will pb.
a) r...
copper will pb.
a) r...
Questions

Mathematics, 21.07.2021 22:40

English, 21.07.2021 22:40

Mathematics, 21.07.2021 22:40


English, 21.07.2021 22:40


Mathematics, 21.07.2021 22:40





Mathematics, 21.07.2021 22:40


Mathematics, 21.07.2021 22:40

Mathematics, 21.07.2021 22:40





History, 21.07.2021 23:20

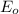 values are known as standard reduction potential values. More the positive reduction potential values, the metal will easily gain electron and will undergo reduction reaction easily.
values are known as standard reduction potential values. More the positive reduction potential values, the metal will easily gain electron and will undergo reduction reaction easily.